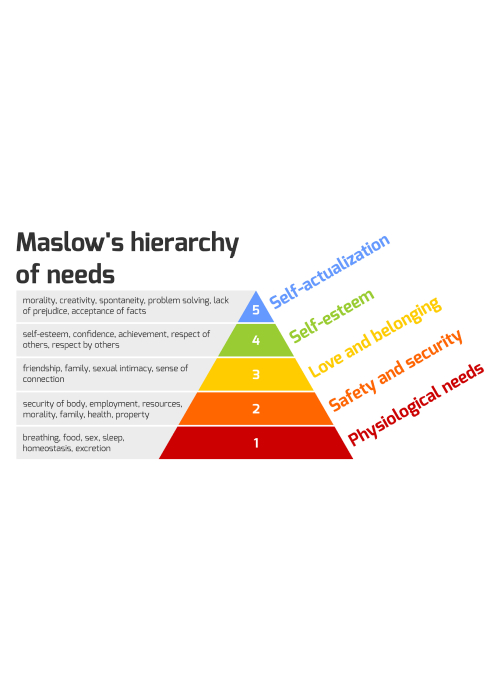
Coronavirus continues to prove it’s here to stay, with cases in all 50 US states and in most countries around the world. Since it has become clear that humans and COVID-19 will coexist for months, if not years, to come, governments and researchers have turned their attention to developing a cure in the form of treatment or a vaccine.
We’ve already talked about our passion for supporting medical research—we do it every day! In that spirit, we’re highlighting potential coronavirus treatments and vaccines from around the world. Check out some of the most promising:
The experimental drug receiving the most initial media attention is remdesivir. Originally created as a potential general antiviral, remdesivir got placed on the fast-track for COVID-19 testing– it’s already being used in five different clinical trials worldwide.
It’s speed in getting to this point can be attributed in part to its role in a past epidemic– it was considered as an option for treating ebola before researchers determined it to be ineffective. As a result, remdesivir has already undergone the initial phases of a clinical trial, and is known to be generally safe for human consumption.
However, the first trial results are already raising concerns and casting doubts on the effectiveness of remdesivir.
Moderna Therapeutics, a biotech firm based out of Cambridge, Mass., is taking a very different approach to their coronavirus vaccine.
According to LiveScience, “Unlike vaccines developed for other viruses, such as measles, this new vaccine does not utilize a weak or dead virus as its base. Instead, the vaccine contains a short segment of genetic material called messenger RNA, or mRNA, generated in a laboratory.”
This new, and potentially groundbreaking, method of vaccine development allows Moderna to fast-track their vaccine. The company is planning to launch clinical trials on healthy patients as soon as April 2020. The new method, though, also casts doubt and uncertainty on how effective the vaccine will prove to be.
China-based Ascletis Pharma Inc.’s is another vaccine ahead of the game. Its potential COVID-19 vaccine is in phase 3, the same phase as Gilead’s remdesivir. Their approach is multi-pronged. It combines two antivirals, one approved to treat HIV and one approved for hepatitis C, that together might effectively treat coronavirus.
The company already reported success with its early-stage clinical trials: 11 patients treated with the combination of treatments were discharged from a hospital in China with positive results.
CanSino, the company responsible for the first effective ebola treatment after the 2014 outbreak, quickly got the green light to begin clinical trials in Wuhan, China for its potential vaccine. Working on a more traditional model, the vaccine would take a piece of coronavirus’ genetic code and combine it with another, harmless virus.
AbbVie is in the process of testing another combination therapy very similar to Ascletis Pharma’s variation. Kaletra® is an existing antiviral drug that treats HIV in adults and children.
AbbVie is working with governments across Europe, North America, Asia, and Australia to treat new coronavirus infections as well as pneumonia in patients.