Join the BEACON-SLE Study
When you consent to contribute your medical records to systemic lupus erythematosus (SLE) research, you join thousands of other patients who have participated in research and empowered their communities through PicnicHealth.
Convenient participation
Earn up to $580 in compensation for your time
18 month study

You may be eligible to join the study if you:
- Are age 18 or older
- Have SLE diagnosed by a rheumatologist
- Started taking Benlysta® (belimumab) in the last six months
- Reside and receive care in the U.S.
What is the BEACON-SLE Study?
The BEACON-SLE Study is a real-world non-interventional study in the United States sponsored by GSK. It follows people with SLE over time to better understand their experiences. The goal of this study is to learn how people living with lupus respond to treatment with Benlysta® (belimumab) in real life.
By allowing PicnicHealth to collect, digitize and de-identify your medical records, you have the opportunity to participate in SLE research while staying in control of your health data.
GSK is sponsoring this study to better understand the real-world experiences of those affected by lupus. Your de-identified data helps inform efforts to develop future treatments and improve care.


What to expect after you consent to join BEACON-SLE

Medical Record Collection

Eligibility Verification

Paid Surveys

Remote Study Visits

In-Person Study Visits
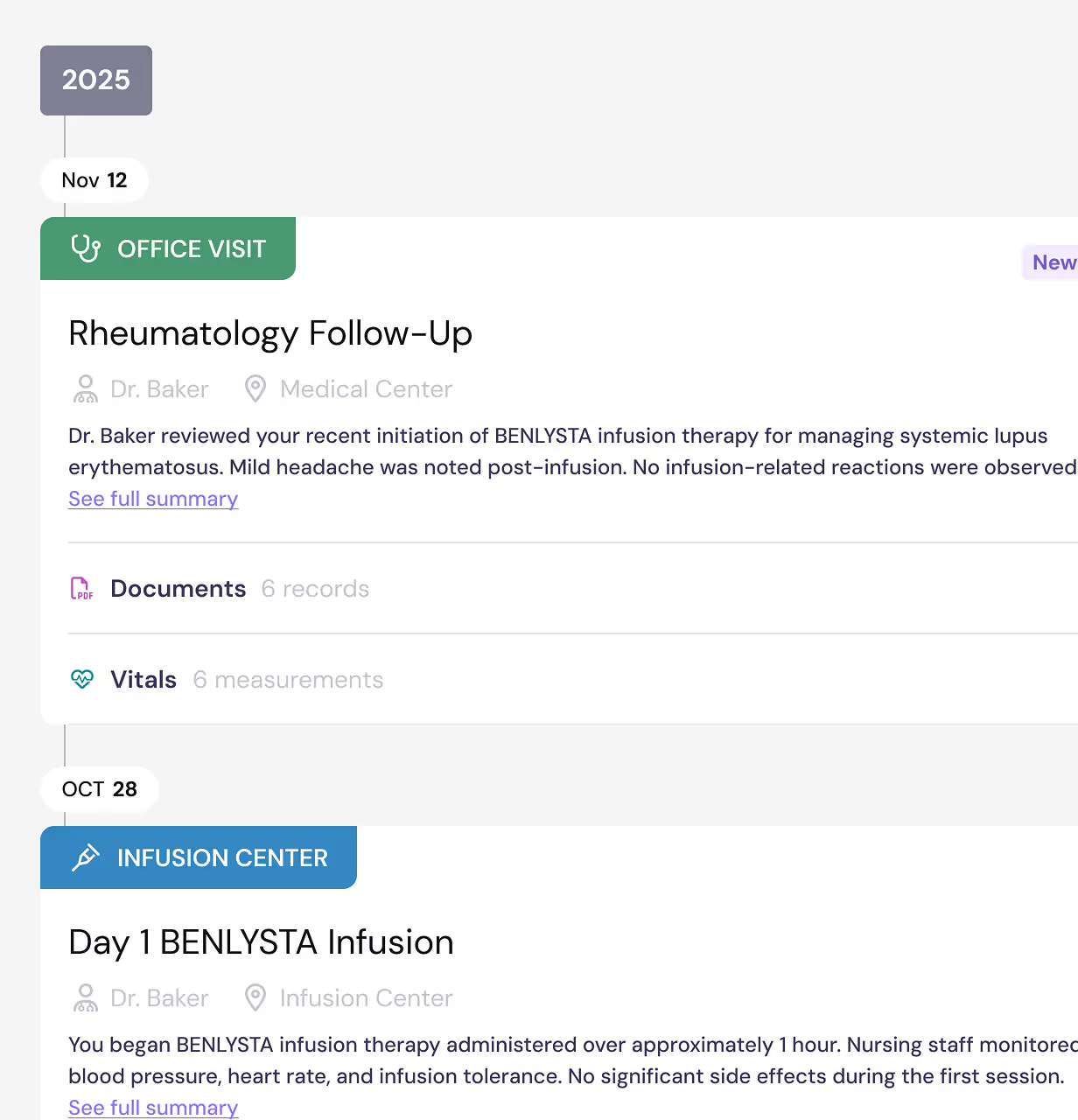
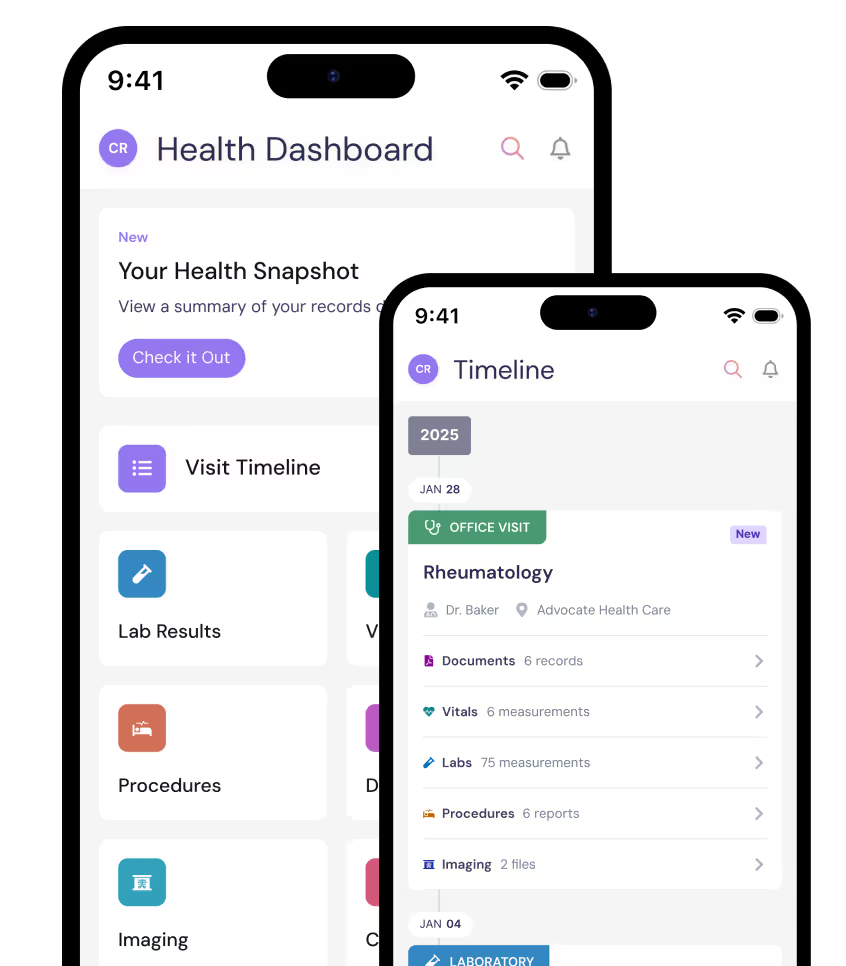
You’ll get a free PicnicHealth account and access to your medical history
By participating in the BEACON-SLE Study, you’ll get access to any of the records we collect. Your medical records are yours to keep, forever.
PicnicHealth Account Benefits
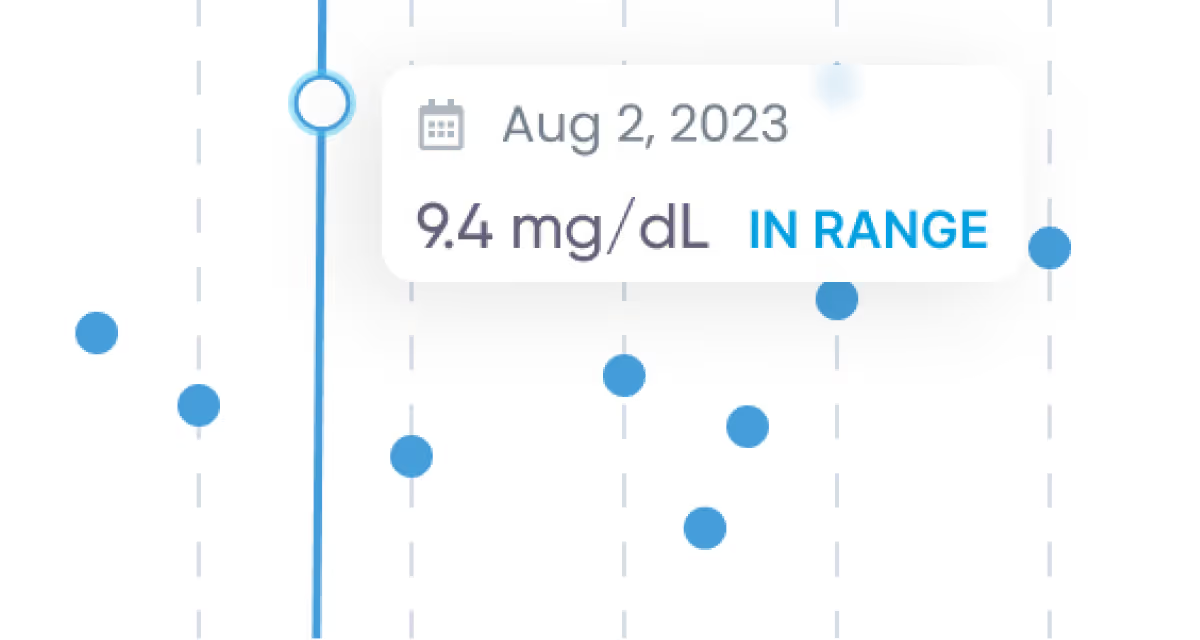
Tools and visualizations
Zoom in on what’s most important and track health trends over time.
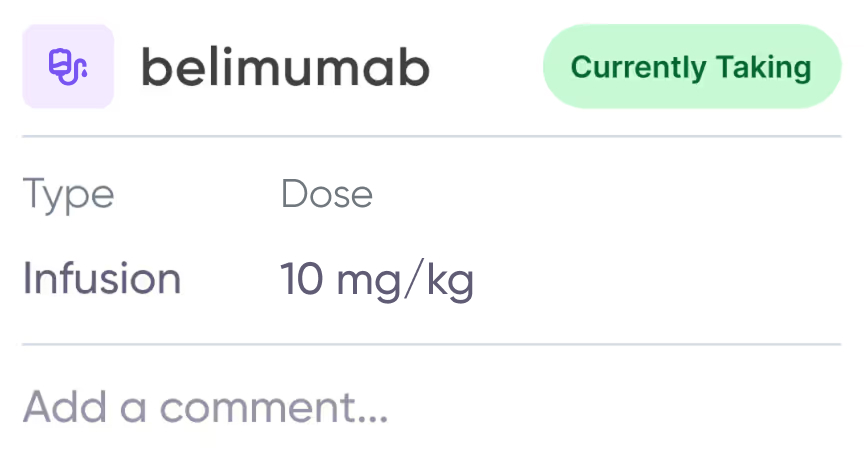
Care management toolkit
Keep track of active medications, care, and health concerns and receive support.

Easy sharing
New doctor? Share your records to get the care you need without wasting time.
Contribute to a deeper understanding of systemic lupus erythematosus and Benlysta® (belimumab)
Sign UpHow It Works
Sign Up in Minutes
To sign up, simply confirm your SLE diagnosis and provide the names of a few healthcare providers. With your consent, PicnicHealth will compile your health history into a secure, regularly-updated digital timeline that only you can access.
Sign Up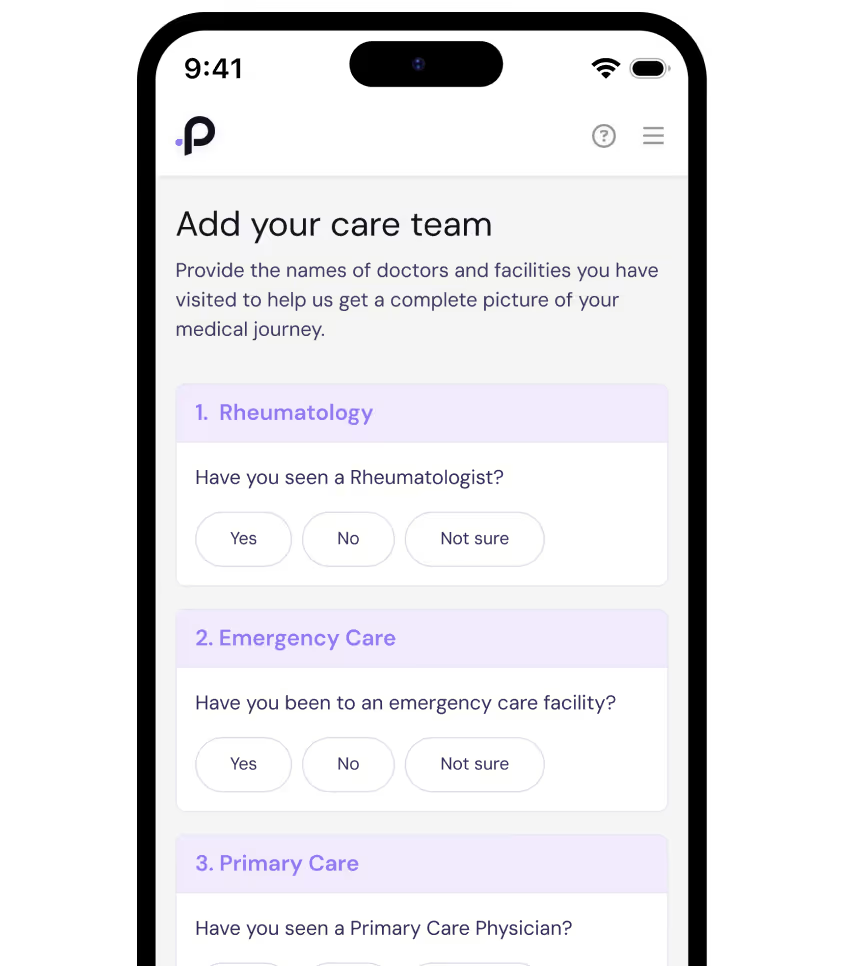
We’ll gather your records
PicnicHealth will do all the work to gather your medical records once you sign up. With secure connections to thousands of healthcare institutions, we’re usually able to find most of your records, and we’ll organize everything into an easy-to-use dashboard.
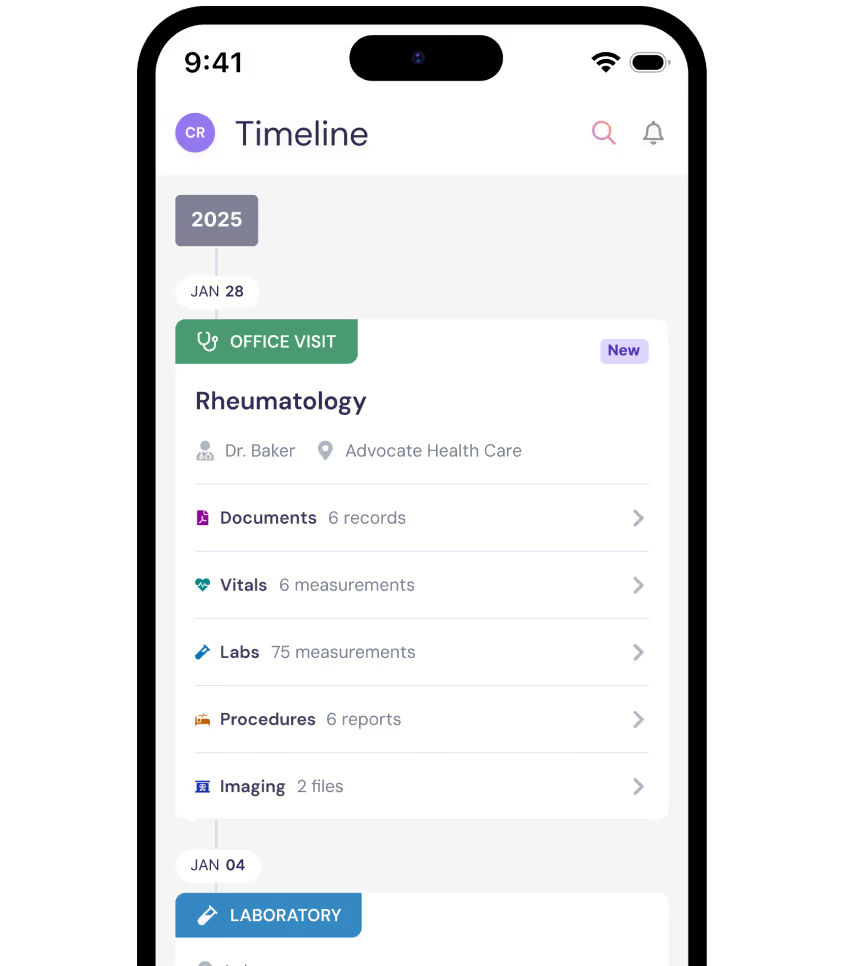
The BEACON-SLE Study combines and analyzes medical records, study visits, and your survey responses
As part of this non-interventional research effort, the BEACON-SLE Study team may analyze your data alongside those of other study participants to gain a clearer picture of how SLE affects daily life. By combining data from many participants, the BEACON-SLE Study team can identify patterns and insights that may not be visible to a single physician.
To protect your privacy, all personal details that could identify you are removed through a process called de-identification, ensuring that researchers receive only the essential health information they need for the BEACON-SLE Study.
Keep your records
We’ll continue to collect new records for the duration of the study. Any records collected are yours to keep forever, to use and share as you please.
Ready to get started?
FAQs
You may be eligible to join the BEACON-SLE study if you:
- Are age 18 or older
- Have SLE diagnosed by a rheumatologist
- Started taking Benlysta® (belimumab) in the last six months
- Reside and receive care in the U.S.
The de-identified data gathered from the surveys, study visits, and medical records is helpful for researchers to understand real world disease and treatment experiences of people with systemic lupus erythematosus (SLE). The BEACON-SLE Study is a real-world non-interventional study in the United States sponsored by GSK. It follows people with SLE over time to better understand their experiences. The goal of this study is to learn how people living with lupus respond to treatment with Benlysta® (belimumab) in real life.
Joining the BEACON-SLE Study is completely optional. If you choose to participate, your medical care, including your Benlysta® treatment will stay the same.
Yes, you can withdraw your consent at any time. This will not affect your medical care or future study participation. When you withdraw, PicnicHealth will stop collecting your records, but any data collected before will remain in the study.
For details about withdrawing your study consent, including details about the surveys and data collection, please reach out to PicnicHealth at hello@picnichealth.com or call (415) 680-3085.
Yes. You will be asked to complete online surveys as part of the study once you sign up and again every three months, which you will receive compensation for every time you complete a survey. It’s important you complete these surveys in a timely manner as they are intended to assess the impact of Benlysta® (belimumab) on your condition.
Yes, virtual study visits are required as a BEACON-SLE Study activity. You’ll be asked to participate in virtual study visits every six months, conducted by video call, where research staff will review your lupus activity using questionnaires.
Additionally, some participants will be approached by their provider to participate in BEACON-SLE. Those participants will be asked to participate in two in-person study visits throughout the study.
No, there is no cost to participate. You will receive PicnicHealth’s service free of charge as part of your participation in the BEACON-SLE Study. If you would like to continue to have PicnicHealth collect your medical records, you may sign-up in the service as a customer at the end of the BEACON-SLE Study or if you are not eligible for the BEACON-SLE Study, you may opt to sign-up for a paid subscription.
You’ll receive $150 upon confirmation of your eligibility. You will also receive up to $100 a year for participating in surveys every three months. Additionally, you will get a free PicnicHealth account with secure access to your medical records - all organized in one place. This account includes tools to help reflect on your care journey.
PicnicHealth plays a key role by collecting and organizing patients’ medical records. For the BEACON-SLE Study, PicnicHealth ensures that relevant health data is accurately captured and made available to the research team in a secure and de-identified format. This enables researchers to analyze real-world outcomes without compromising your privacy.
PicnicHealth is a leading health technology company simplifying healthcare for everyone and a partner selected by GSK to support the study. Through a combination of clinical expertise and technology, PicnicHealth provides patients with their organized medical records to prepare for doctor visits and advocate for themselves. PicnicHealth will only share de-identified data with the research team — meaning your name and personal information are removed before researchers access anything.
GSK is the pharmaceutical company manufacturing Benlysta® (belimumab). PicnicHealth, in turn, is the data partner for the BEACON-SLE Study, facilitating the secure collection and de-identification of health records for GSK’s research purposes. Both parties collaborate to ensure a smooth and compliant study process.
Once you enroll and provide authorization, PicnicHealth begins by searching for your records through our connected medical record network. If necessary, direct requests can also be sent to your healthcare providers to retrieve copies of your medical records. Some electronic records can be available within an hour, while records from individual providers may take a couple of weeks. Overall, this process can take a few weeks depending on how quickly providers respond. PicnicHealth will continuously update your timeline as new records are received and processed.
The BEACON-SLE Study is not a clinical trial. A traditional clinical trial involves testing a specific treatment, like a drug or medical device. The BEACON-SLE Study will only review de-identified survey answers, study visits, and your medical records produced during your visits to the doctor.
PicnicHealth will never share your records without your explicit consent. By signing up for this study, you're agreeing to share your de-identified data with GSK for the purpose of the BEACON-SLE Study. No one outside PicnicHealth - including your doctor or insurance company - will be able to see your medical records unless you choose to share them by clicking "Send Records" at the top of the PicnicHealth timeline within the app. You will be the only person who can access your medical records on PicnicHealth.
GSK, as a study sponsor, is conducting BEACON‑SLE to better understand how Benlysta® (belimumab) helps people impacted by SLE and their treatment journey to include clinical outcomes and quality of life in the real-world setting — information that can help patients, doctors, and health systems make better decisions.
PicnicHealth follows Health Insurance Portability and Accountability Act (HIPAA)-compliant practices and uses advanced end-to-end encryption and security measures, the same technology banks use to keep information secure. Your records are only shared with your permission.
For more details or assistance regarding the PicnicHealth platform and medical record services, visit our website or reach out to the PicnicHealth team at hello@picnichealth.com or call at 415-680-3085.

“With PicnicHealth, I was able to eliminate the tedious work and stress of managing my records allowing me to focus on my care and living!”
PicnicHealth Research Participant

